Our Science
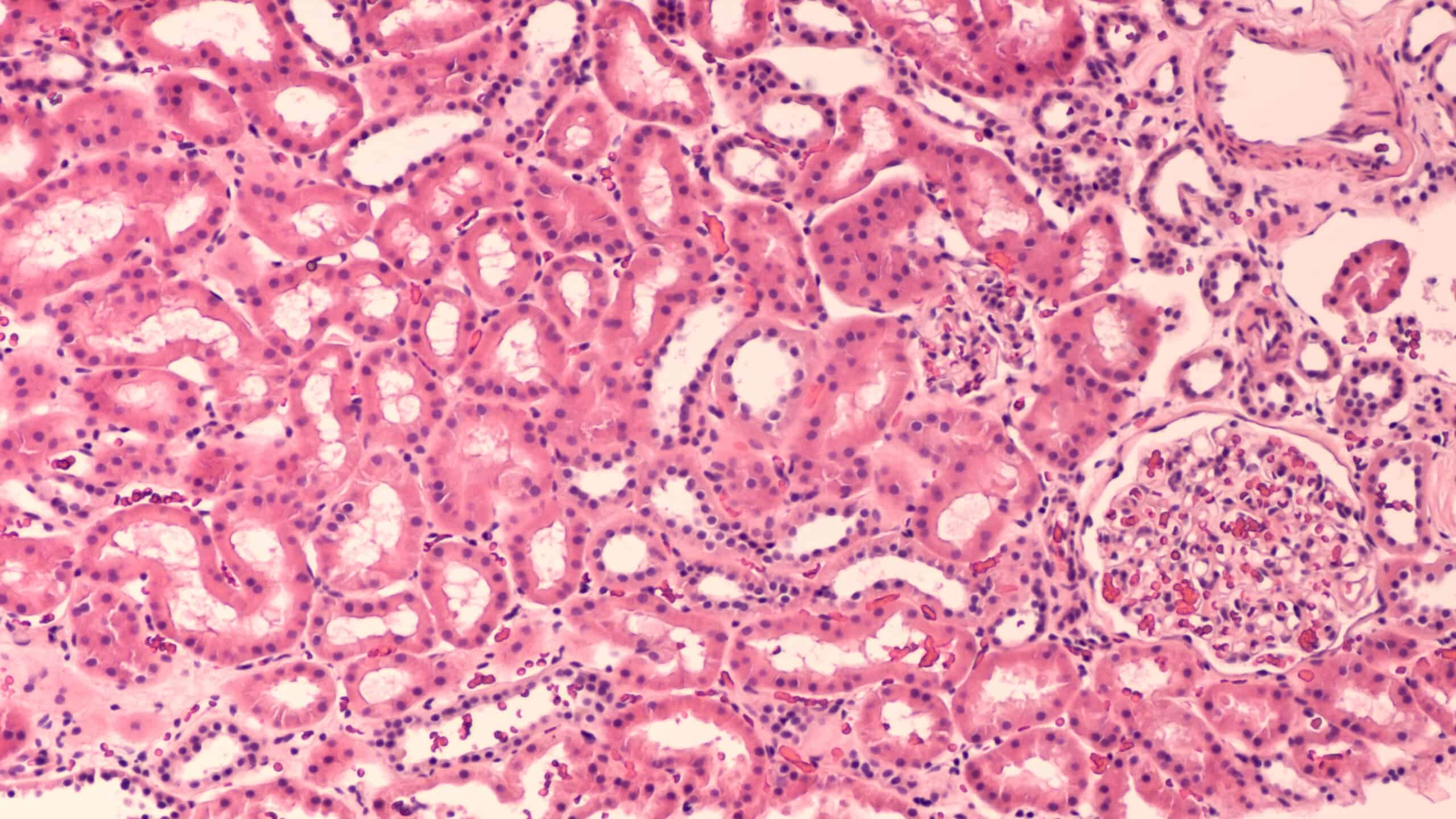
Integrins
From the family of 24 integrins found in vertebrates, a subset of eight recognise an arginine-glycine-aspartic acid (RGD) sequence in the latency associated peptide (LAP) that maintains transforming growth factor-beta (TGF-β) in an inactive form. TGF-β is a key component in the activation of fibroblasts, the cells responsible for collagen deposition and the build-up of scar tissue associated with fibrotic disease. Whilst direct inhibition of TGF-β has been associated with undesired side-effects, TGF-β levels can be modulated by inhibition of specific integrins. RGD-integrins are able to activate TGF-β by binding to the LAP, which makes the selective inhibition of RGD-integrins an attractive therapeutic strategy.
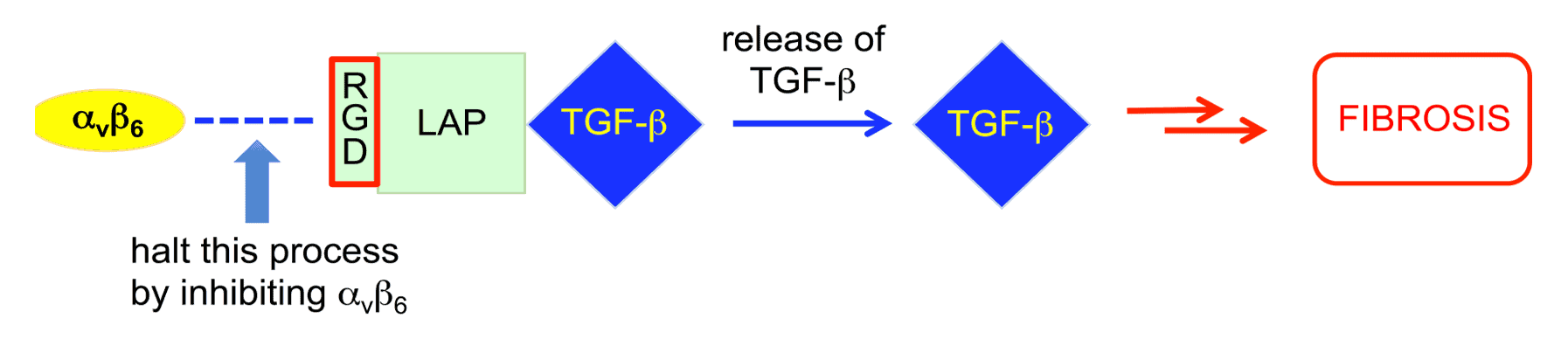
Our approach to the inhibition of RGD-integrins is to mimic the RGD sequence in the LAP binding site by designing a molecule with a basic site, a linker and an acid group to mimic the arginine, glycine and aspartic acid residues respectively.
We have designed and synthesised a substantial library of small molecules with various selectivity across distinct chemical series, with drug-like physicochemical properties for inhaled, oral or intravenous administration.
Therapeutic applications
Our priority is to develop an inhaled treatment for idiopathic pulmonary fibrosis (IPF), a chronic interstitial lung disease, characterised by excessive scarring of the lungs. IPF is invariably fatal, with median survival rates of between two and four years following diagnosis.
Expression of the αvβ6 integrin is elevated in fibrotic lung tissue compared to healthy tissue, supporting its status as a therapeutic target. Subsequently, we will develop Alevin’s pipeline to address other unmet medical needs including chronic kidney disease (CKD), acute kidney injury (AKI) and cancer.
Highlights from the Scientific Founders’ Publications
Pharmacological characterisation of GSK3335103, an oral αvβ6 integrin small molecule RGD-mimetic inhibitor for the treatment of fibrotic disease. European Journal of Pharmacology, 2021.
Late-stage functionalization via Chan-Lam amination: rapid access to potent and selective integrin inhibitors. Chemistry A European Journal, 2020.
Translational pharmacology of an inhaled small molecule αvβ6 integrin inhibitor for idiopathic pulmonary fibrosis. Nature Communications, 2020.
Structure activity relationships αv Integrin antagonists for pulmonary fibrosis by variation in arylsubstituents. ACS Medicinal Chemistry Letters, 2014.
For an expanded list of relevant publications from the Scientific Founders, please click here.